Synthesis of Fluorescent Five- and Six-Membered Ring
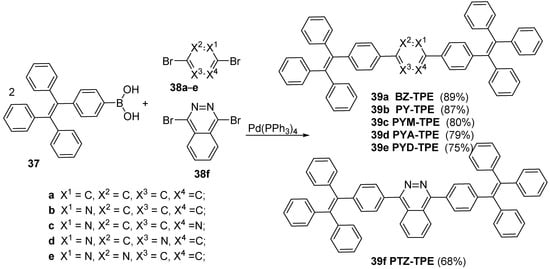
The synthesis of fluorescent azaheterocycles continues to arouse strong interest due to their great potential for application as sensors and biosensors, luminophores on in the construction of Organic Light-Emitting Diode (OLED) devices, laser and other semiconductor devices, as well as to their potential biological properties as antimicrobial, antifungal, anticancer, antituberculosis antioxidant and anti-HIV agents. The advantages of the azaheterocyclic fluorophores, such as small size, enriched photostability, a wide and tunable spectral range, and, frequently, high brightness, are the reason why these fluorophores are preferred and used in various medical application. Probe structure can be modified to adjust excitation and emission wavelengths, target-binding affinity, chemical reactivity, and subcellular localization.

S-Heterocycle Synthesis

Structure and docking of diazines in the tubulin binding site: (a

Frontiers Chemical insights into the synthetic chemistry of five-membered saturated heterocycles—a transition metal–catalyzed approach

PDF) A Review on the Synthesis of Fluorescent Five- and Six-Membered Ring Azaheterocycles

Scheme 2. Synthesis reaction of tetracyclic azide derivatives 4a-d

Chemical reporters for biological discovery. - Abstract - Europe PMC

Synthesis of FRET probes 5 and 6 incorporating a five-membered (m = 0)

Synthesis and characterization of 2,8-diazaperylene-1,3,7,9-tetraone, a new anthracene diimide containing six-membered imide rings.

Molecules, Free Full-Text
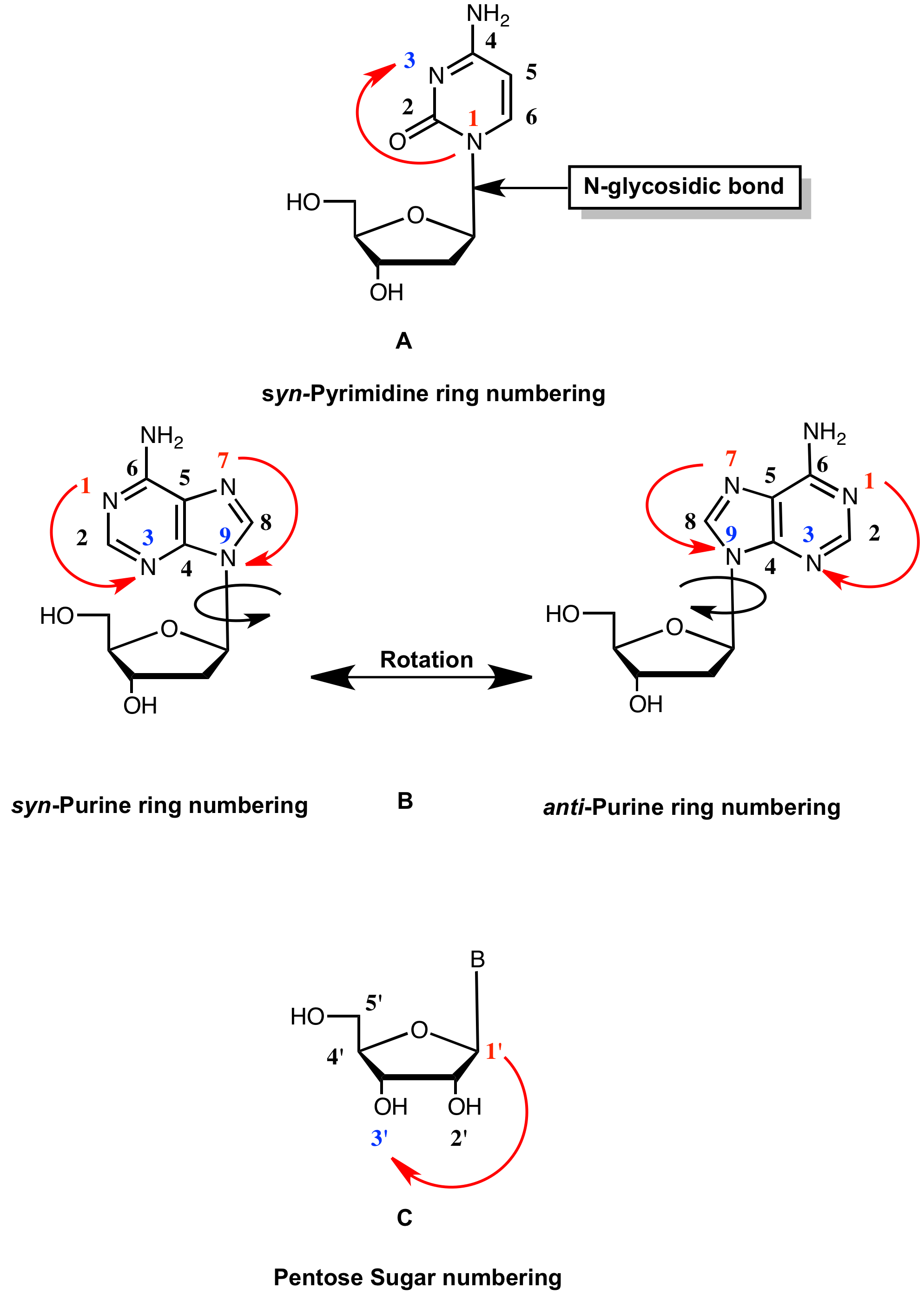
Glen Report 33-15: Technical Note - DNA and RNA Nucleoside Numbering System
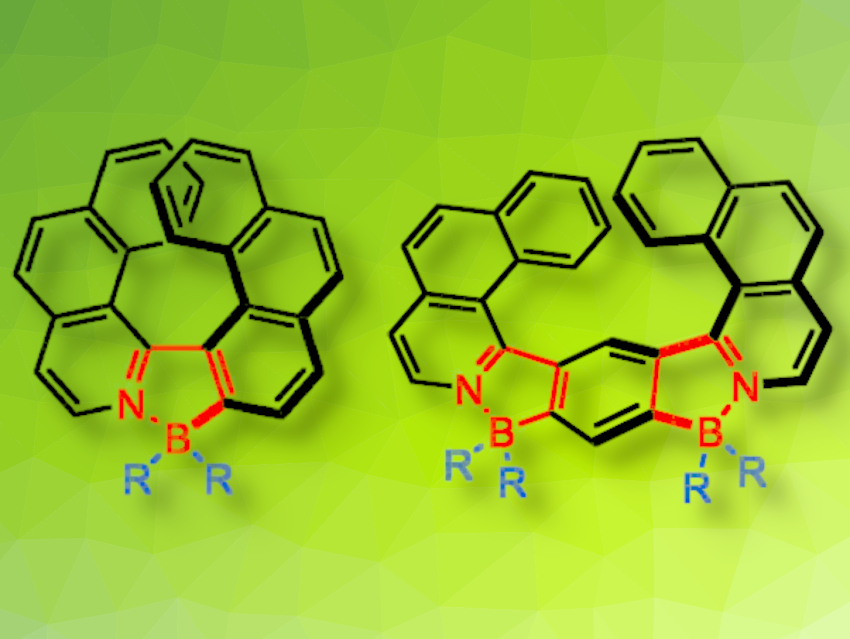
Boron-Containing Helicenes - ChemistryViews

Processes, Free Full-Text

Results of the in vitro growth inhibition (GI%) caused by