1) 2 23 g ethanol is dissolved in 36 g water. Find mole fraction

Click here:point_up_2:to get an answer to your question :writing_hand:1 223 g ethanol is dissolved in 36 g water find mole fraction of ethanol2
Click here👆to get an answer to your question ✍️ -1- 2 23 g ethanol is dissolved in 36 g water- Find mole fraction of ethanol -2- 0-5 -3- 0-2 -4- 0-8 TIN

23 g ethanol is dissolved in 36 g water. Find mole fraction of

SOLVED: 23 gram ethanol is dissolved in 36 gram water. Find mole

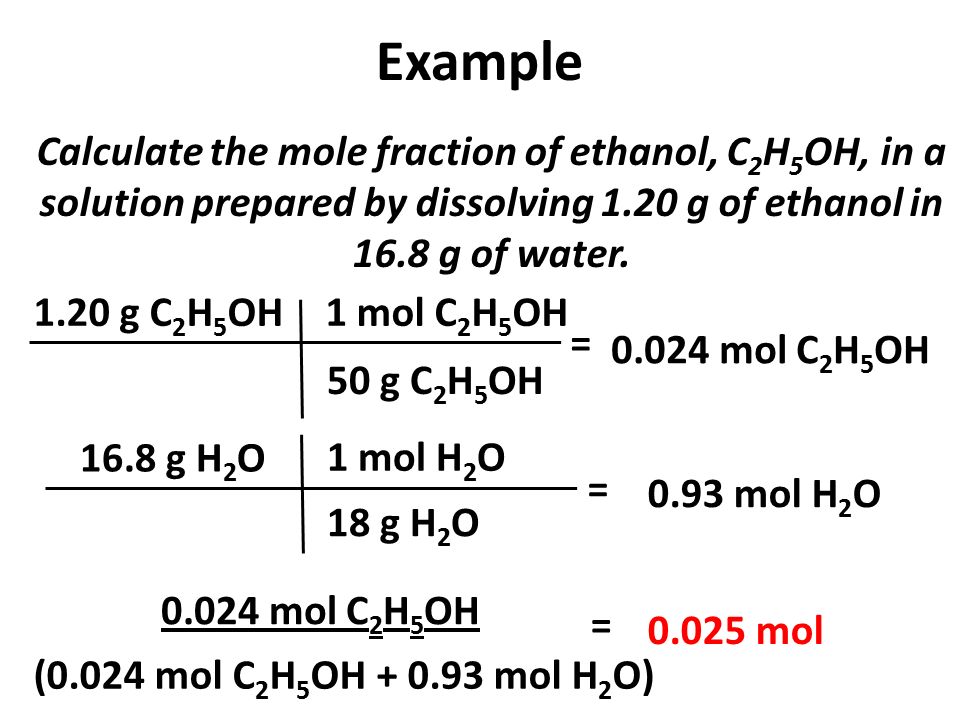
Calculate the mole fraction of ethyl alcohol and water in a
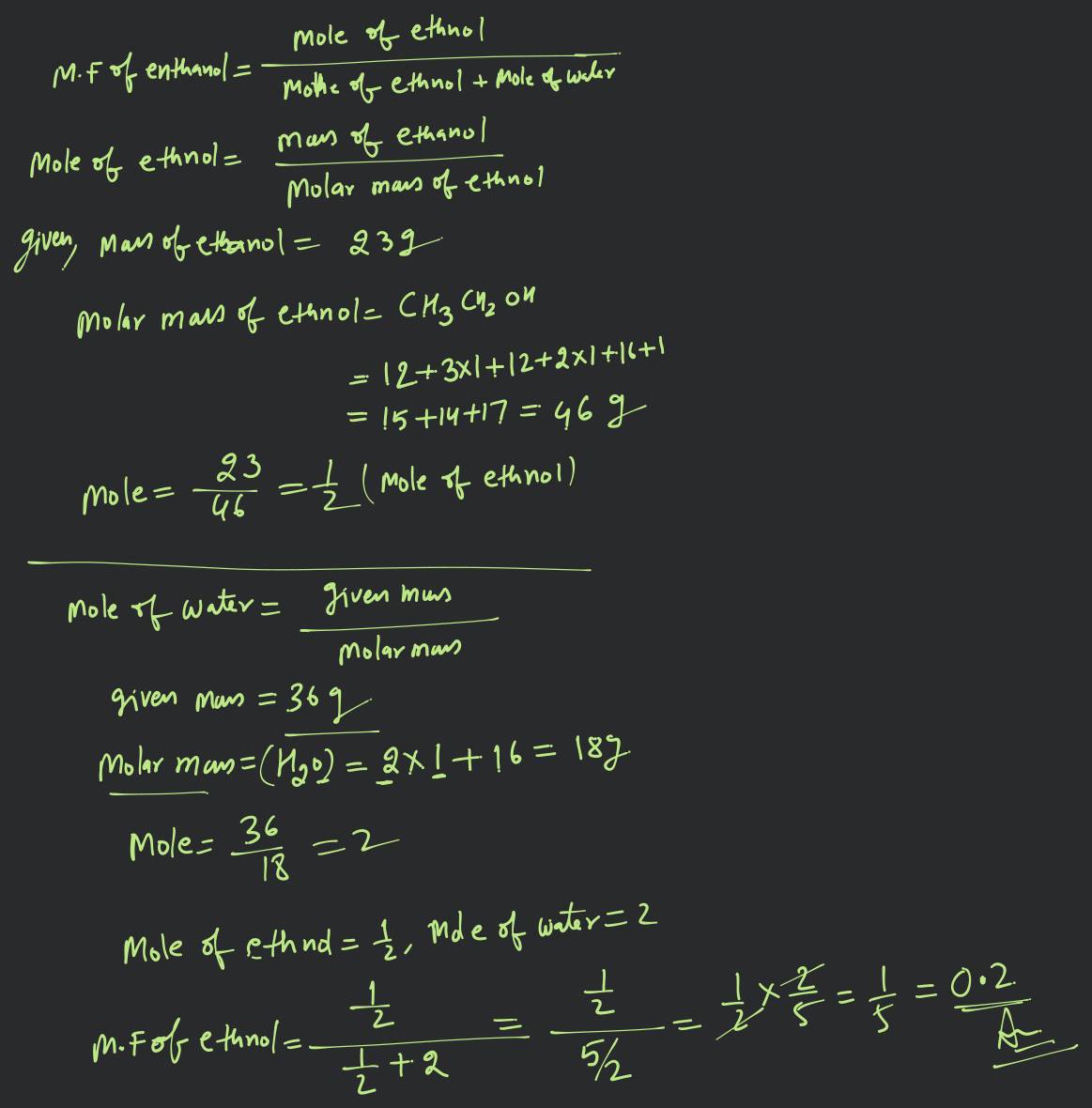
23 g ethanol is dissolved in 36 g water. Find mole fraction of

Calculate the molality of a solution of Ethanol (C_2H_5OH) in

Calculate mole fraction of ethyl alcohol and water in a solution
Exam Review #2b. Introduction to Stoichiometry What is Mole Ratio

23 g Ethanol is dissolved in 36 g water. Find mole fraction ethanol

SOLVED: 23 gram ethanol is dissolved in 36 gram water. Find mole

1) 2 23 g ethanol is dissolved in 36 g water. Find mole fraction

SOLVED: Question 6 of 37 A solution is made using 126.1 g of water