Compressibility factor Z = PV / nRT is plotted against pressure as

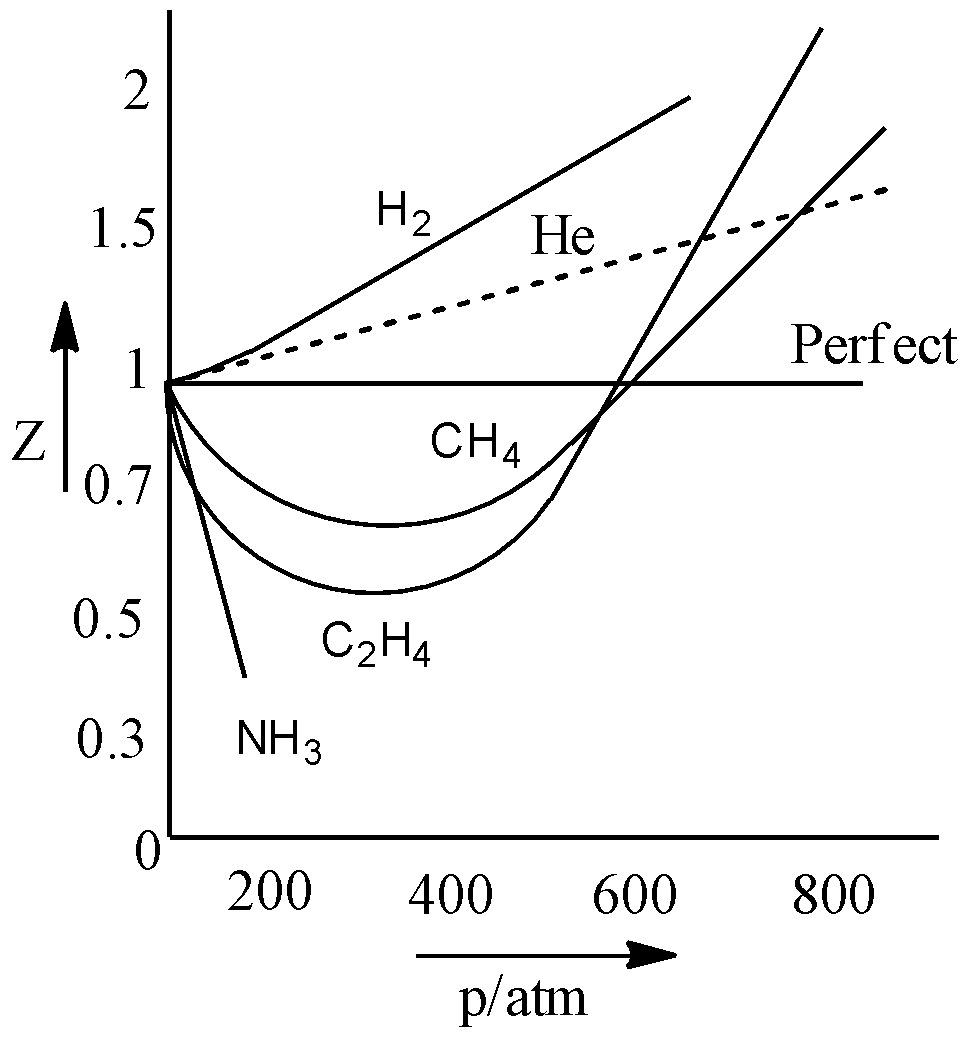
Compressibility factor Z = PV / nRT is plotted against pressure as shown below:What is the correct order for the liquefiability of the gases shown in the above graph? A. CO 2
Compressibility factor Z - PV - nRT is plotted against pressure as shown below-What is the correct order for the liquefiability of the gases shown in the above graph- A- CO 2- CH 4- N 2- H 2B- H 2- CH 4- N 2- CO 2C- CH 4- H 2- N 2- CO 2D- H 2- N 2- CH 4- CO 2

1.5 Real Gases and the Virial Equation - Mail

1.1: Thermodynamic Variables and Equations of State - Chemistry LibreTexts

Compressibility factor Z - Gaseous State

Gas Compressibility - an overview

Compressibility factor of water vapor along its saturation curve. Error

Compressibility factor (Z=(PV)/(nRT)) is plotted against pressure

Compressibility factor (Z=(PV)/(nRT)) is plotted against pressure

PV Compressibility factor Z= nRT is plotted against pressure : N. Ideal gas What is the correct order of liquefiability of the gases shown in the above graph? H
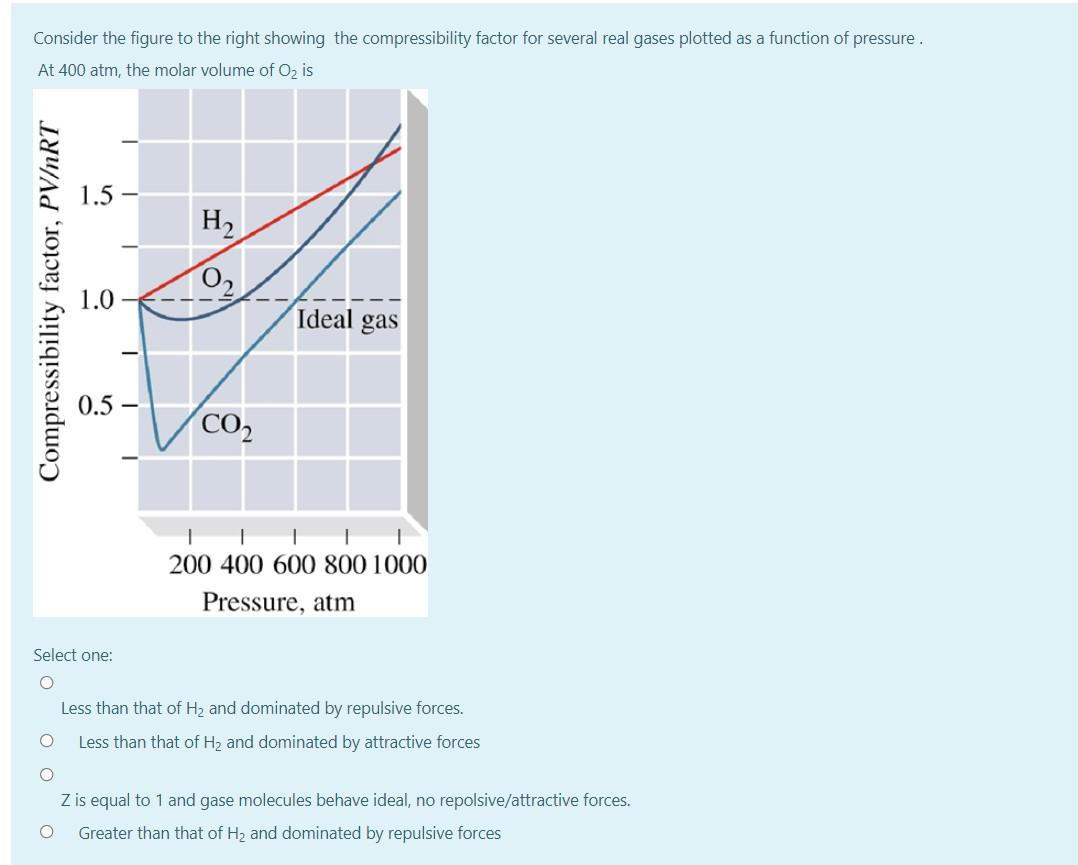
Solved Consider the figure to the right showing the

Compressibility factor - Wikipedia

Thermodynamics Exam 1 (Ch.1-3) Defs. Flashcards

Gas compressibility factor Z: Ideal gas vs Real gas
The given graph represents the variations of compressibility factor `Z=PV// nRT` vs `
The answer to this question is a single digit integer, ranging from 0 to 9.The compressibility factor Z for an ideal gas will be________.