Ideal–Universal Gas Law

Definition: The Universal or Ideal Gas Law describes the relationship between all four properties (pressure, volume, number of moles, and temperature) as well as a gas constant called “R.” NOTE: The Ideal Gas Constant “R” has constant a value of 0.0821 L.atm/mol.K Relation: The relation between pressure (P) volume (V), number of moles (n) and…
How to calculate the characteristic gas constant of a gas (air) - Quora

Avogadro's Law

Let's Review Regents: Chemistry--Physical Setting Revised Edition by Albert S. Tarendash M.S., Paperback

Regents Chemistry--Physical Setting Power Pack Revised Edition by Albert S. Tarendash (Ebook) - Read free for 30 days

Ideal Gas Law — Overview & Calculations - Expii
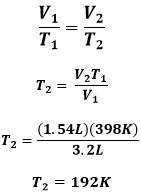
Charles' Law

Bohr's Model of Atom and Equation
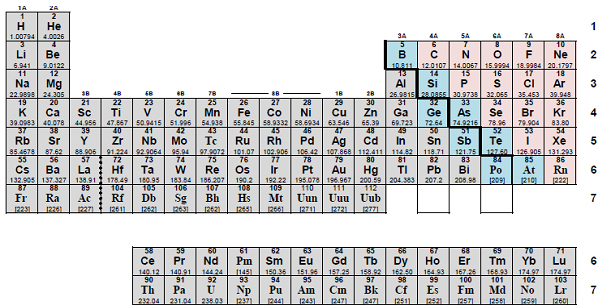
Anatomy of the Periodic Table

Ideal Gas Law Formula and Examples

Combined Gas Law - Definition, Formula, Examples

The Ideal Gas Law, Equation & Constant
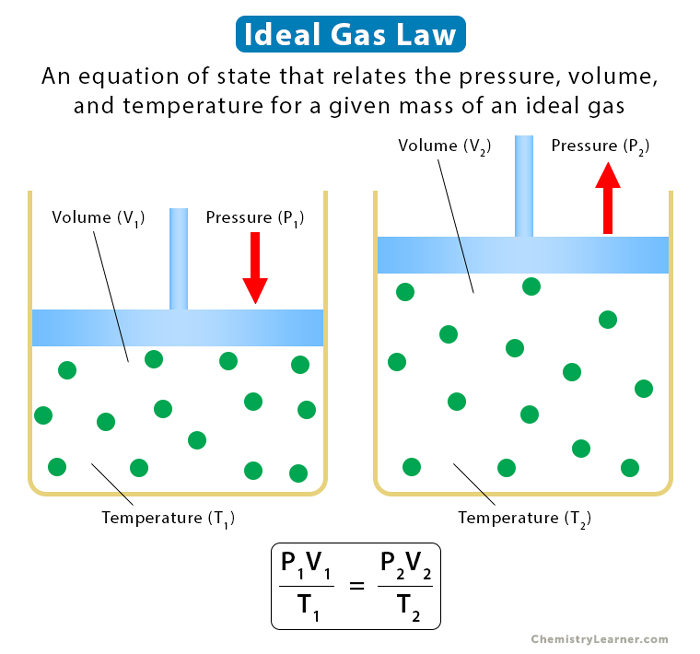
Ideal Gas Law: Statement, Characteristics, Formula & Problems