FDA has new mammogram guidelines for dense breast disclosure. What
/cloudfront-us-east-1.images.arcpublishing.com/pmn/EMEDJI4AQVBZJNJHKLTY2ZSPJY.jpg)
In Pennsylvania, senators unanimously voted in favor of a bill to fund genetic testing to women at higher risk of breast cancer.
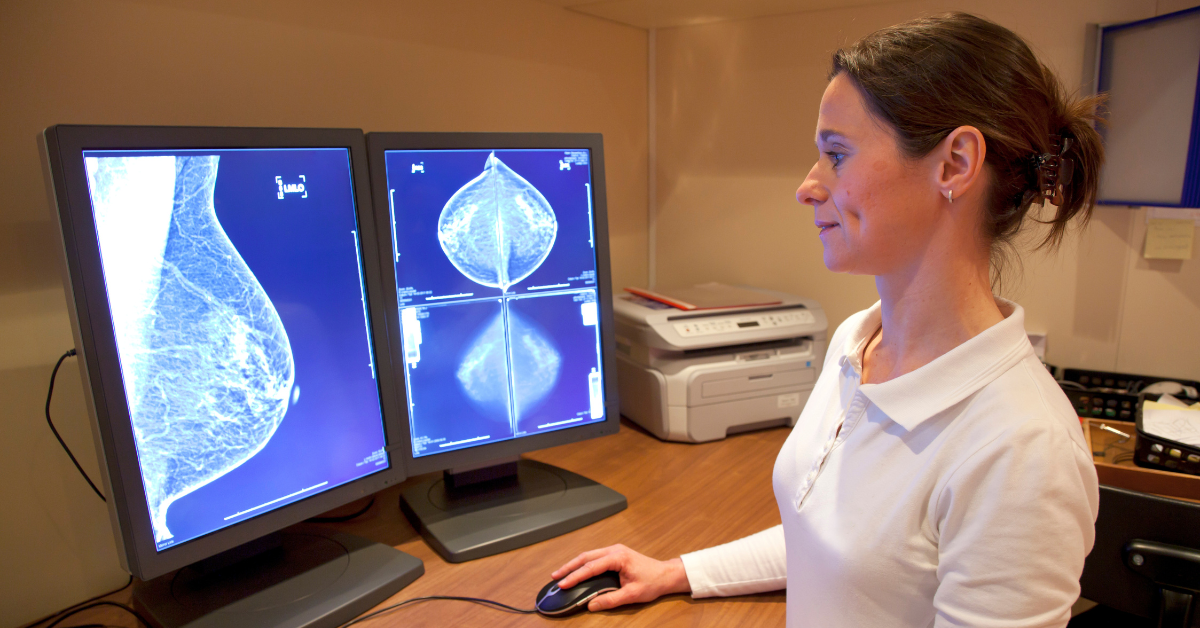
New FDA guidelines on breast density notifications and

U.S. proposes letters telling women dense breasts make it harder

FDA to Implement New Mammogram Regulations to Support Women with
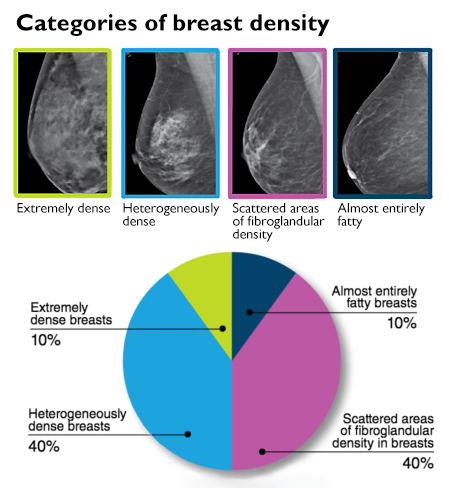
FDA aims to level field on breast-density disclosure to patients
Breast cancer screening: FDA proposes mammogram changes

A Review of Breast Density Implications and Breast Cancer

Rad Tech CE, ASRT, ARRT® CE, Category A Credits

Secondary Screening With ABUS Improves Cancer Detection in Dense

FDA mammography update raises cost questions
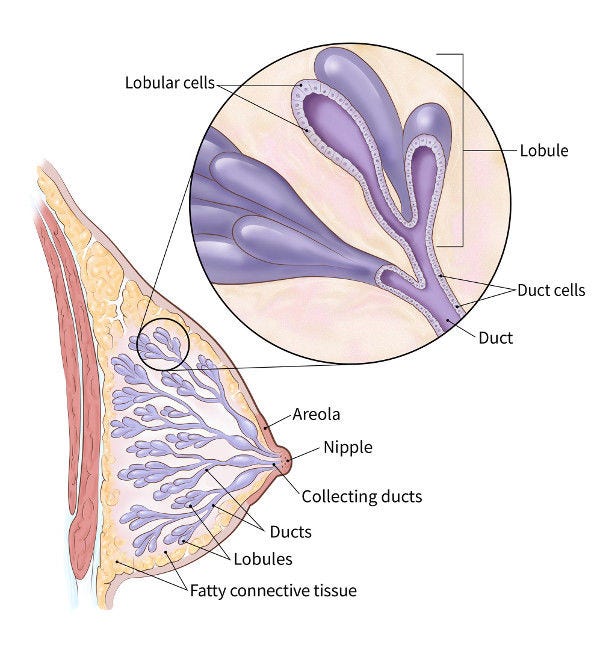
Dense Breast Tissue Breast Density and Mammogram Reports

Many Women with Dense Breasts May Not Need Additional Screening - NCI