Aβ(1-42) tetramer and octamer structures reveal edge conductivity
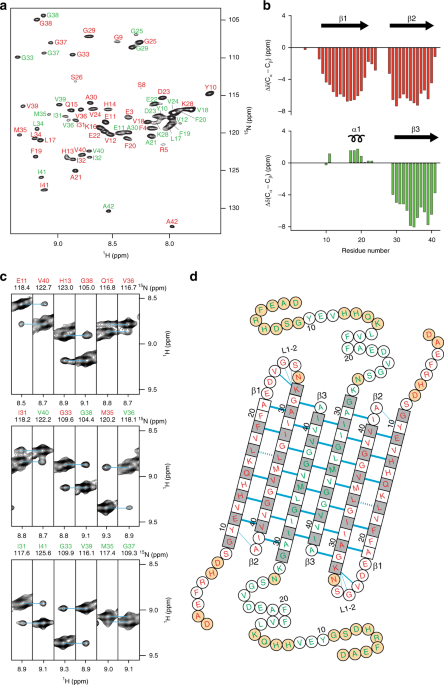

A glimpse into the structural properties of α‐synuclein oligomers
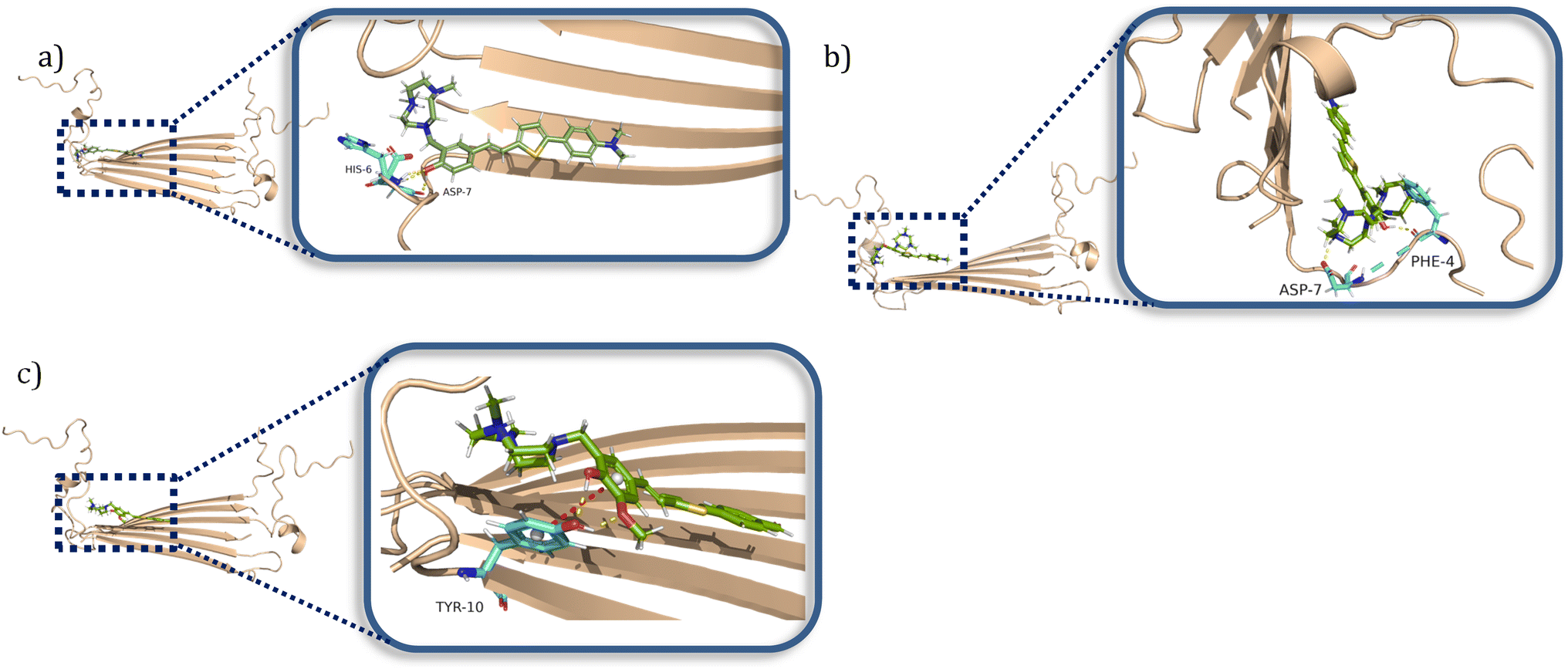
Amphiphilic stilbene derivatives attenuate the neurotoxicity of soluble Aβ 42 oligomers by controlling their interactions with cell membranes - Chemical Science (RSC Publishing) DOI:10.1039/D2SC02654F

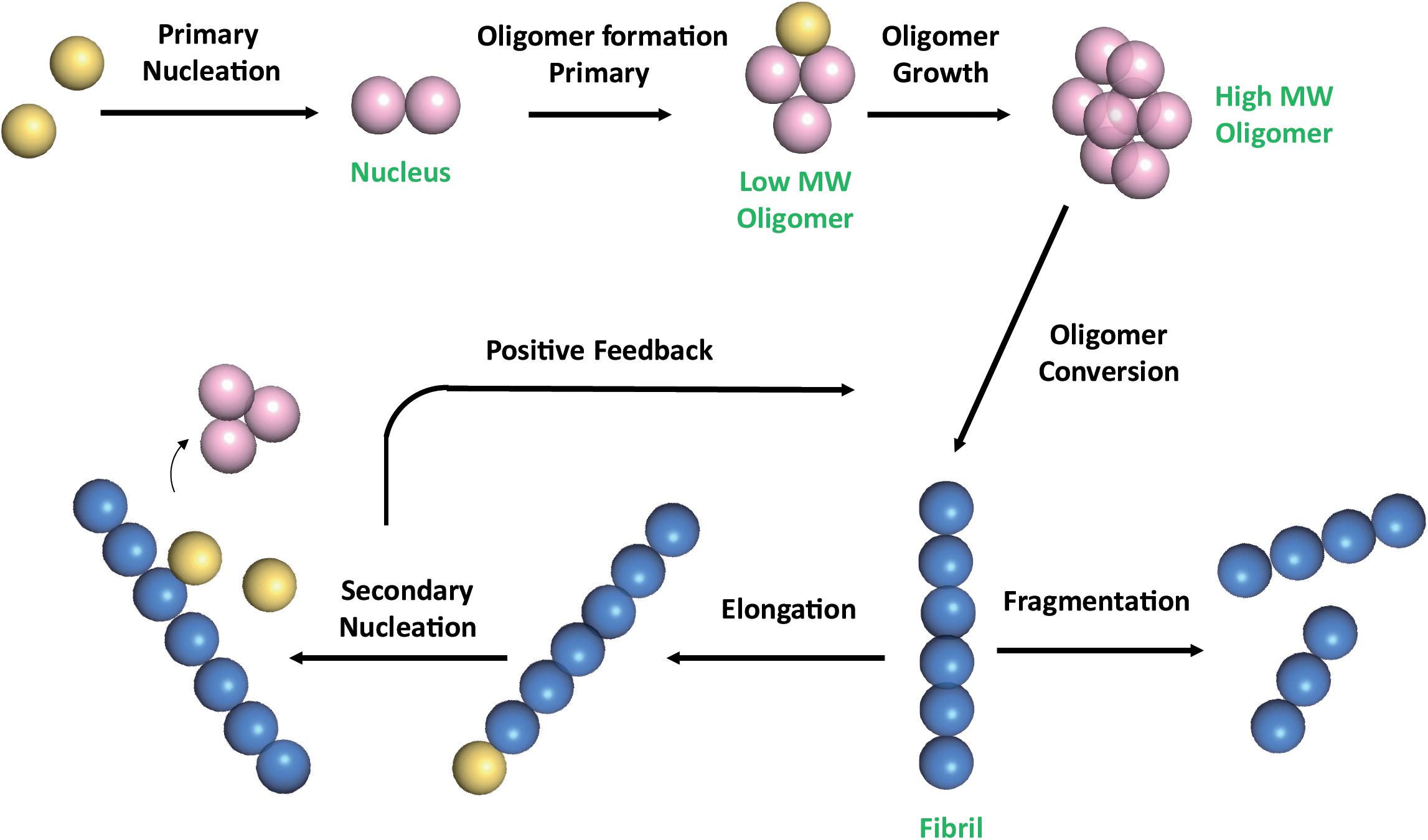
The amyloid concentric β-barrel hypothesis: Models of amyloid beta 42 oligomers and annular protofibrils

Aβ(1-42) tetramer and octamer structures reveal edge conductivity pores as a mechanism for membrane damage
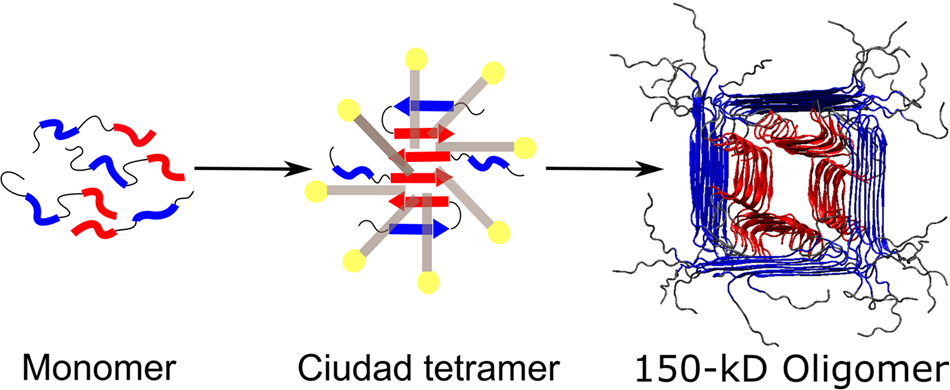
A common pathway for detergent-assisted oligomerization of Aβ42

PDF) Aβ(1-42) tetramer and octamer structures reveal edge pores as a mechanism for membrane damage
Frontiers Natural Compounds as Inhibitors of Aβ Peptide Aggregation: Chemical Requirements and Molecular Mechanisms

Effect of lipid saturation on amyloid-beta peptide partitioning and aggregation in neuronal membranes: molecular dynamics simulations
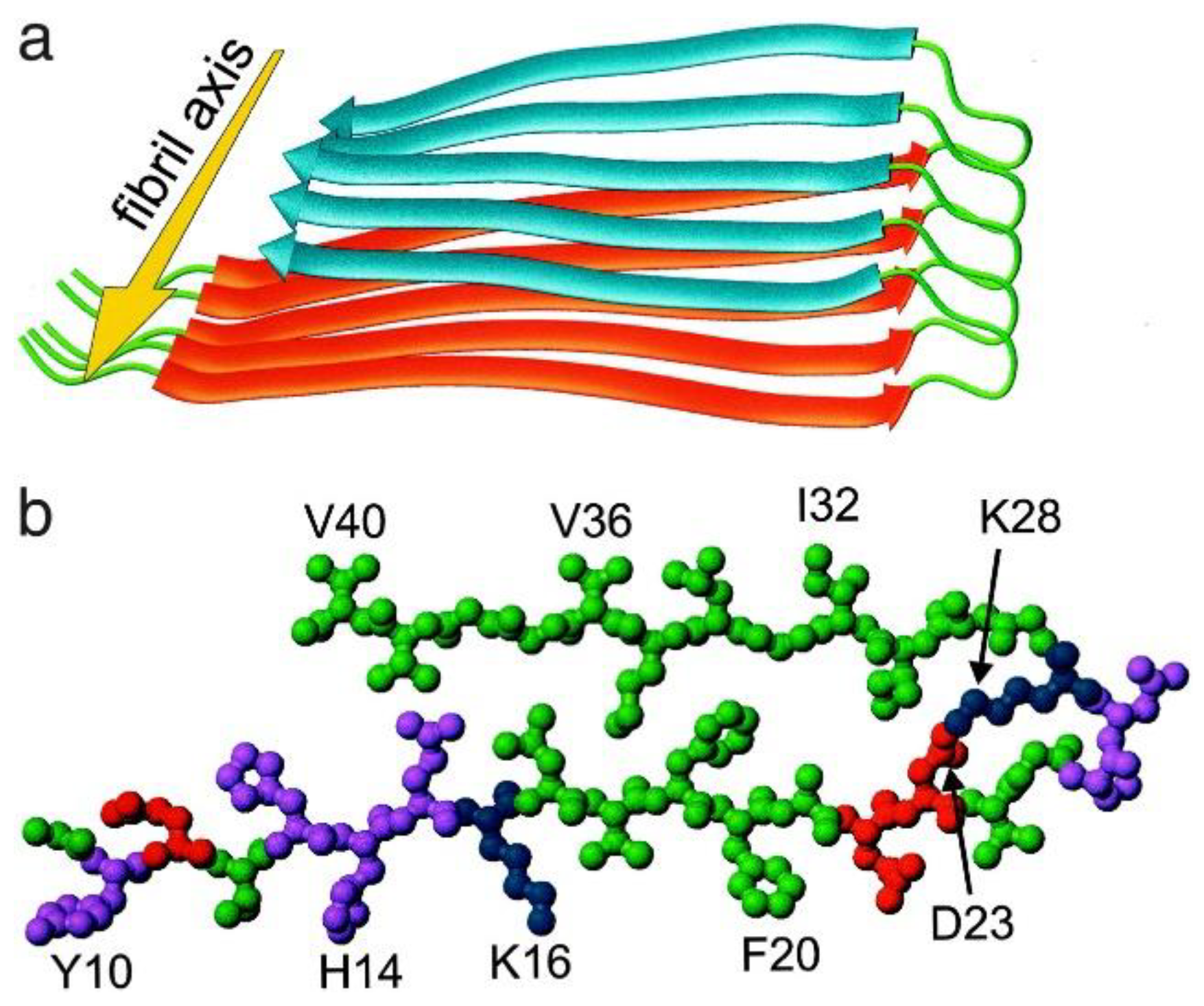
Molecules, Free Full-Text

Molecular dynamics simulations reveal the importance of amyloid-beta oligomer β-sheet edge conformations in membrane permeabilization - ScienceDirect

Ultrasensitive electrochemical detection of amyloid-beta oligomers using double amplification strategy by MXene substrate and covalent organic framework-based probe,Talanta - X-MOL

PDF] Alzheimer´s Disease-associated Aβ42 Peptide: Expression and Purification for NMR Structural Studies